Hello, my name is Rebecca Jones, and I’m about to begin the final year of an undergraduate Biochemical Engineering, M.Eng., degree at University College London (UCL).
Last summer, I interned in the Biochemical Engineering department at UCL, working with Ultra-Scale-Down (USD) devices that had been designed and created at UCL. One such device was the USD shear device. Normally, the effect of shear on separation steps, such as centrifugation, is extremely time-consuming and uneconomic to evaluate at large-scale. The USD shear device offers a way of evaluating the effect of shear on downstream unit operations with only millilitre quantities of precious product. In large-scale processes, shear stress can be applied to the process fluid, especially in areas such as the feed zone of a centrifuge. This shearing effect can break open suspended cells, making it more difficult for them to be separated from the surrounding liquid in the centrifuge. Shearing can also degrade the product by exposing it to harmful intracellular enzymes.
Further devices that I worked with included the USD membrane device and USD formulation device. It was my job to learn about these devices and their operation in order to create detailed scientific manuals. Further to this, I also created instructive videos for the operation of the devices. These were to be used predominantly by students, so they could become familiar with the devices before being introduced to them in practical sessions. Filming also gave me the opportunity to show aspects of the device that students wouldn’t normally get to see. A video showing the dismantling and inside of the pilot-scale centrifuge was shot. This internship gave me great experience producing scientific documents and self-managing projects.
As an SRF Summer Scholar under the mentorship of Dr. Ivan Wall at UCL, I will be studying the translational challenges faced by tissue-engineered hollow organs. I am so happy to be researching bioengineered organs. The potential to transform a patient’s quality of life by alleviating the need for immunosuppression or reliance on organ donors is immense and it is my real motivation for wanting to explore this field.
Manufacturing Requirements for Translating Hollow Organ Tissue Engineering to Routine Clinical Practice
My summer project in the Wall lab involves a systematic review of the key translational challenges faced by tissue-engineered hollow organs with a particular emphasis on the current bioprocessing bottlenecks that need to be addressed in order to gain widespread clinical adoption. Clinical success of transplanting bioengineered hollow organs has already been achieved: bioengineered tracheas being the most prominent example. These have been transplanted using both natural scaffolds that have been stripped of their host cells and artificial scaffolds. However, although many different approaches have been taken to create viable bioengineered organs, there is currently no consensus as to the ‘gold standard’ that should be adopted in order to manufacture these products.
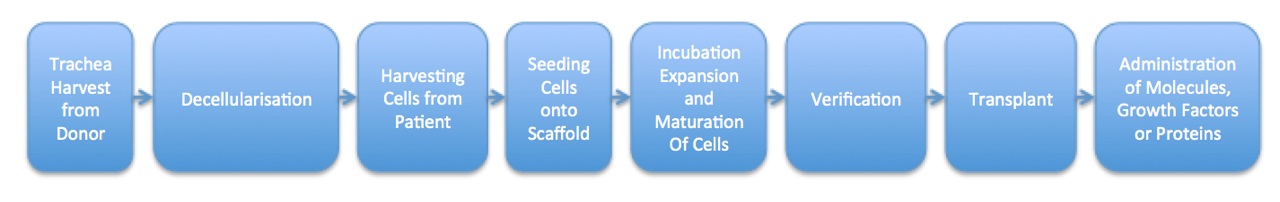
Figure 1. Sample Process Map for the Manufacture and Delivery of a Hollow Bioengineered Organ.
This process map shows the manufacturing and delivery steps involved in the transplantation of a hollow bioengineered organ, such as a trachea. Steps include harvest of a donor trachea, delivery of the new organ to the patient, and administration of any bioactive molecules after the surgery.
This summer, I shall be breaking down each stage of the process needed to construct these bioengineered products: from harvesting/creating the scaffold to reseeding it with recipient cells to transplanting the organ and administering bioactive molecules. At each stage, I shall look at the potential problems with current approaches and the implications of those problems on widespread manufacturing and clinical adoption with the goal of offering a possible solution. I shall also look at engineering parameters, e.g. temperature and pH, at each stage in the production process. I hope to detail any desirable process control parameters that have been assessed and to identify those that may have been overlooked.
The project will also entail interviewing key international academic and industrial leaders in order to identify the greatest translational challenges faced by the organ bioengineering community. My interviews will particularly focus on the product-specific barriers that relate to bioengineered airways with the goal of identifying the best standardized production methods that are currently available. My project will also identify future improvements that are likely to come to fruition and explain how they will affect manufacturing scale-up and the chain of custody from patient to bioprocess to clinical implantation. In summary, I aim to produce a review article that will provide practical process engineering solutions for a more informed long-term biomanufacturing strategy for a range of tissue-engineered products, such as bioengineered tracheas.
Future Plans:
I intend to study and gain my Master’s in Biochemical Engineering this year. As my main Master’s Project, I want to work in a lab that is researching bioengineered organs. I would also like to gain more insight into the clinical implications of this research.


